Research Outline
We integrate a wide variety of molecular (nano)-blocks such as organic molecules, polymers, biomolecules, photofunctional molecules, metal complexes, inorganic compounds, metal nanocrystals depending on the purpose and investigate causal relationships between the self-assembled nanostructures and their functions. Based on the supramolecular design that we have cultivated, we are now developing and researching molecular systems with advanced photo-functionalities.
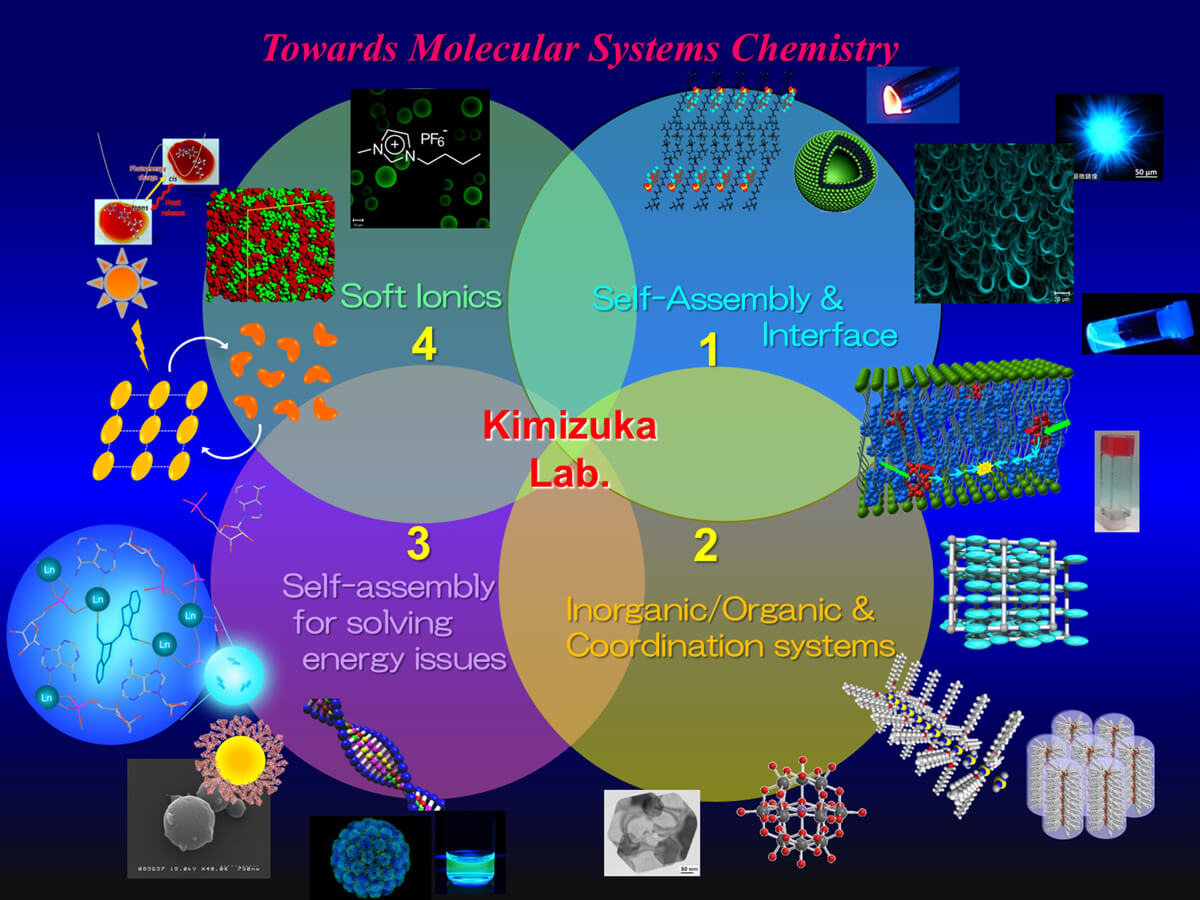
Figure 2 Research areas for the study of “Molecular Systems Chemistry” in Kimizuka Laboratory –
1. Molecular self-assembly phenomena and functions in solution, condensed phase and at interfaces.
2. Interdisciplinary research areas of inorganic and organic chemistry: coordination chemistry, coordination polymers, supramolecular polymers, metal clusters, metal nanostructures and plasmonics.
3. Fusion of molecular self-assembly and photochemistry for solving energy and environmental problems.
4. Soft-Ionics: Development of functional molecular assemblies directed by electrostatic interactions, self-assembly in ionic liquids, and interfacial materials chemistry which maximize the full potential of ionic liquids.
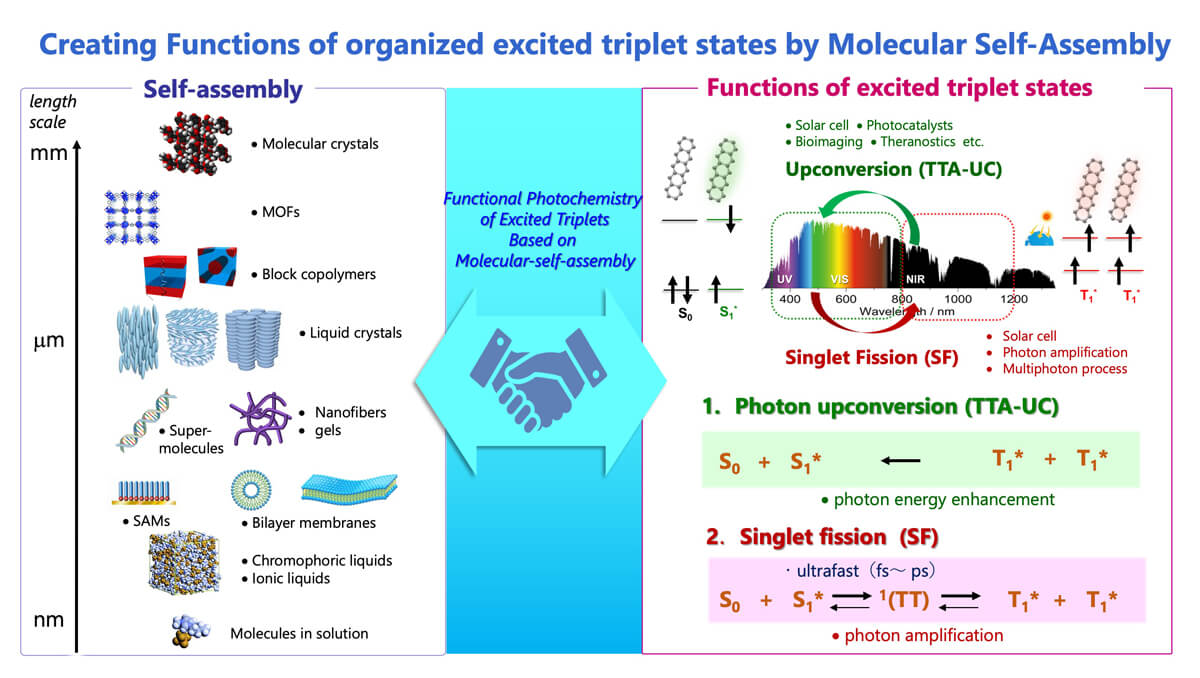
Towards “Molecular Systems’ Chemistry” – Self-assembled Photon Upconversion systems
In biomolecular systems such as thylakoid membranes and mitochondrial inner membranes, static self-assembly phenomena and thermodynamically uphill processes such as ATP synthesis are dynamically conjugated. This molecular system’s function in life is difficult to realize by extension of conventional molecular self-assembly or supramolecular chemistry governed by the “thermodynamic energy minimum principle.” In other words, in addition to structure formation by self-assembly, a new perspective of molecular systems chemistry is needed.
We define “Molecular Systems Chemistry” as a Chemistry for temporally and spatially conjugating physical and chemical phenomena to create useful work based on self-assembly.1 To link self-assembly to useful work, it is necessary to have techniques for molecular-level control of the energy landscapes, including their ground, excited, and transition states, based on the molecular organization of multiple functional components. To realize this, we have started research on “photon upconversion using molecular self-assembly.
Photon upconversion via triplet-triplet annihilation (TTA) (TTA-UC) has attracted much attention as a mechanism that allows upconversion to be observed using relatively low-intensity excitation light at the solar level. In this TTA-UC, a donor (sensitizer) and an acceptor (emitter) are used in pairs. First, the donor absorbs light and enters a triplet excited state, and then the triplet energy is transferred to the acceptor. When two acceptor molecules in the excited triplet diffuse into the solution and collide, one of them enters the excited singlet state, which is higher than the triplet state and emits high-energy light from this excited singlet state (Fig. 3).
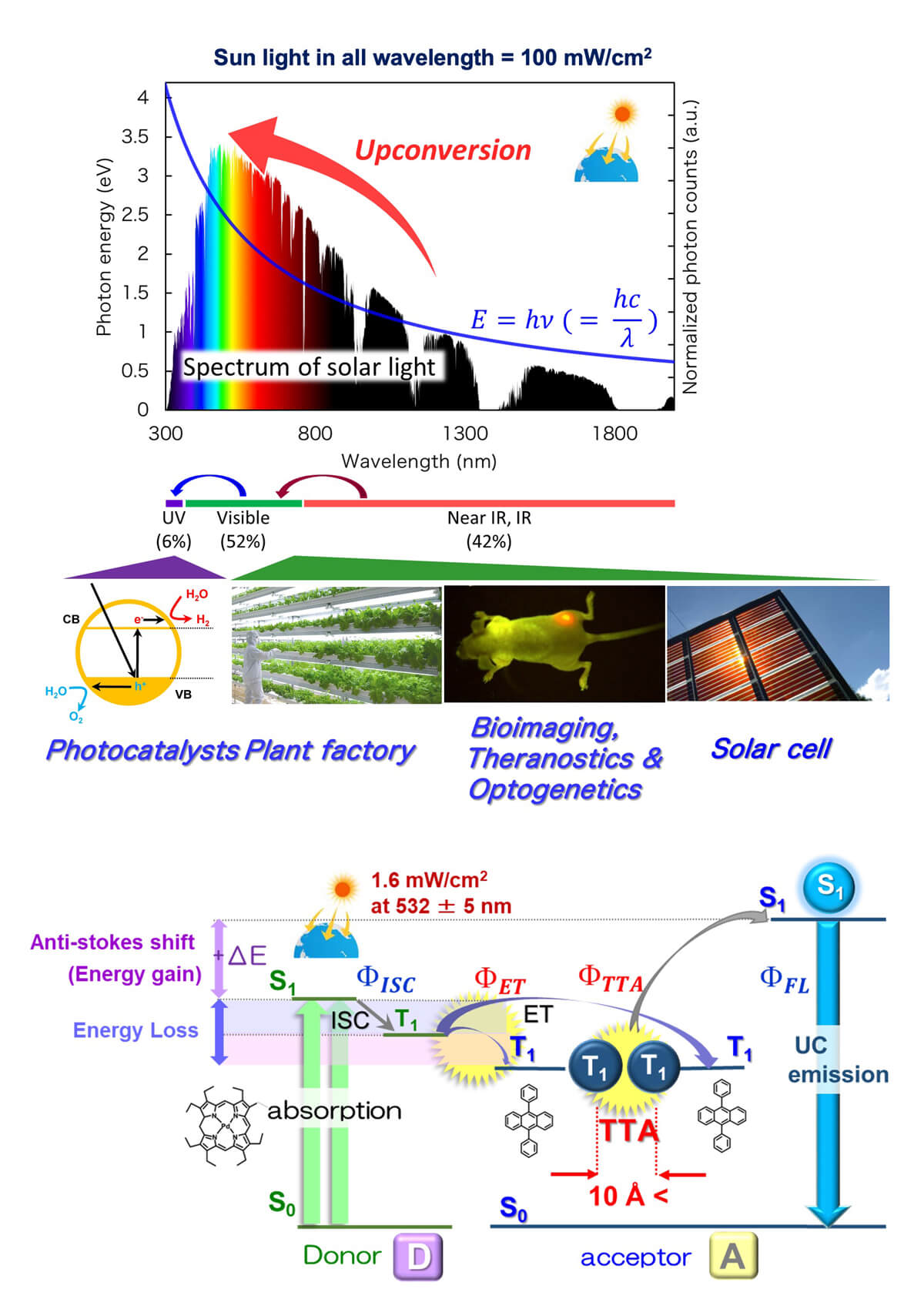
Figure 3. Photon upconversion based on triplet-triplet annihilation (TTA-UC).¹
In previous studies, donors and acceptors were dispersed in solutions or polymers, and energy transfer was induced by diffusion and collision of dye molecules (Fig.4 left). However, molecular diffusion is limited in viscous media such as in polymer matrices, which is important for applications. Moreover, the excited triplet state is deactivated by oxygen, which requires the system to be fully deaerated.
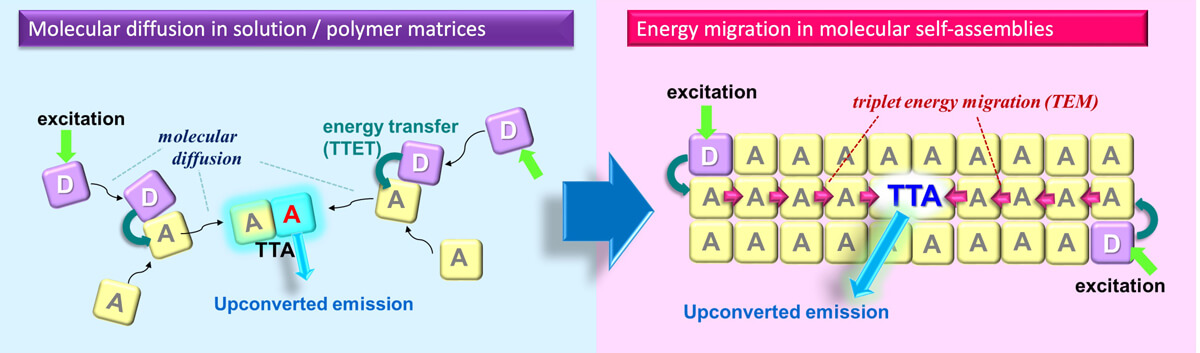
Figure 4. left: conventional TTA-UC based on molecular diffusion phenomena,
right: TTA-UC based on triplet energy migration in designed molecular self-assemblies (our work). ¹ ²
In our previous study, we have investigated chromophore-containing molecular self-assemblies that showed efficient singlet energy transfer via energy migration (Fig.5). As far as we are aware, this is the first report of singlet energy migration in molecularly self-assembled gels (supramolecular gels).
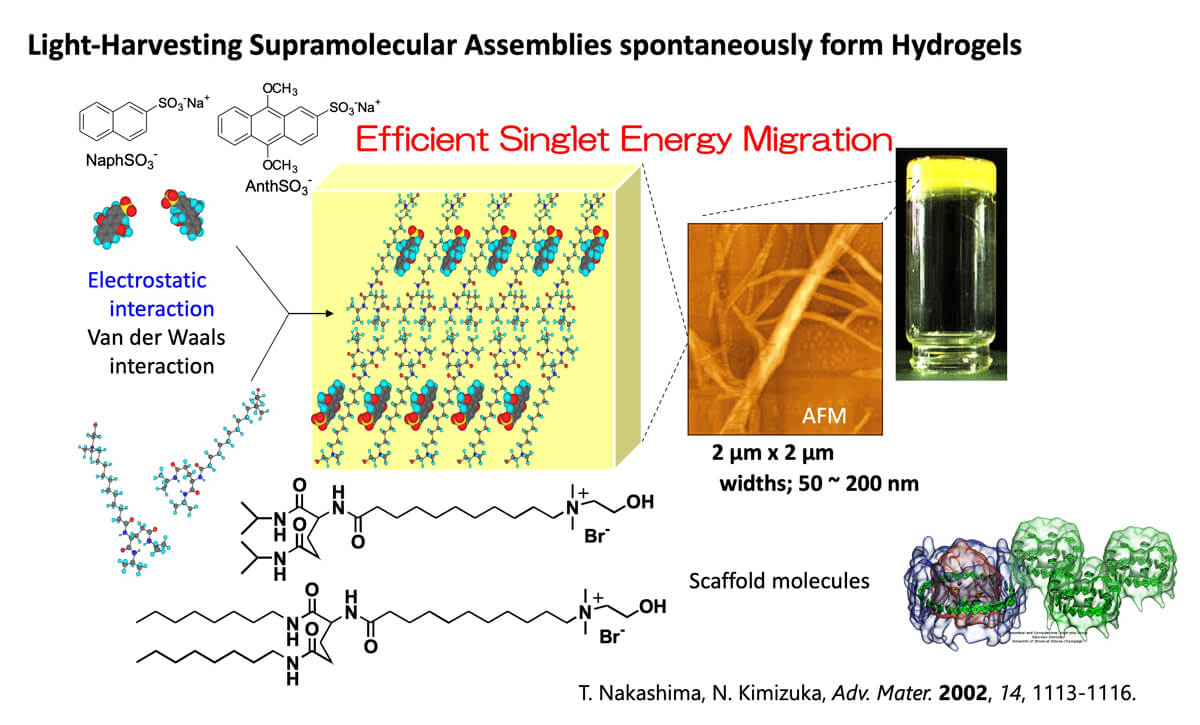
Figure 5. Singlet energy migration in light-harvesting supramolecular hydrogels.³
We also found that excited triplets of platinum porphyrins are protected from dissolved oxygen by inclusion into coordination network nanoparticles (Fig.6). It clearly indicated that the designed self-assembly allows us opportunities to handle excited triplets in an air-saturated environments.
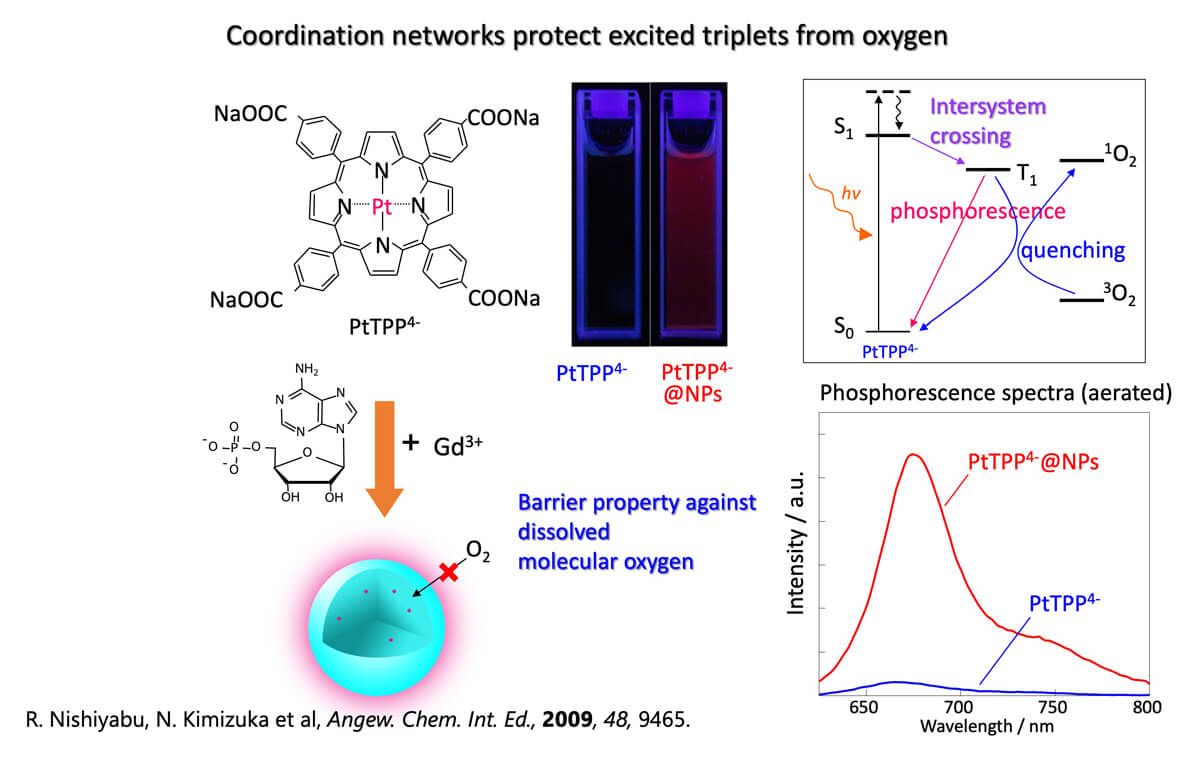
Figure 6. Protecting excited triplet state of Pt Porphirins by coordination networks. ⁴
Based on these findings, we have developed photon upconversion molecular systems based on triplet energy migration (diffusion of triplet excited states) between chromophore molecules in condensed or self-assembled systems (Figure 4, right). We have developed various molecular systems that show efficient photon upconversion properties via controlling energy landscapes, which led to new developments in supramolecular chemistry, fundamentals of molecular systems chemistry, and the development of novel up-conversion systems. (Figure 7).1, 2
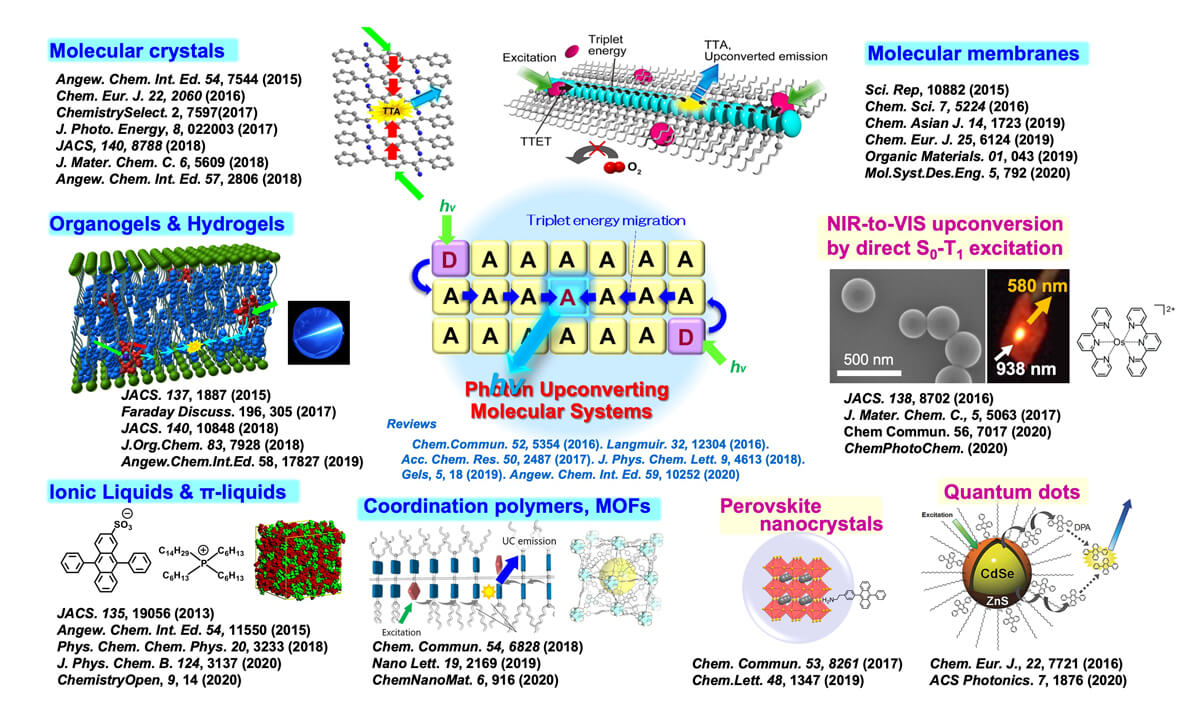
Figure 7. Photon upconversion based on triplet energy migration in controlled molecular self-assembly and newly developed photon upconversion systems.
Publications Relevant to the Project
1. N. Kimizuka, N. Yanai, M-a. Morikawa, Langmuir, 2016, 32, 12304-12322. Invited Feature Article
2. N. Yanai, N. Kimizuka, Chem. Commun., 2016, 52, 5354-5370. Feature Article
3. T. Nakashima, N. Kimizuka, Adv. Mater, 2002, 14, 1113-1116.
4. R. Nishiyabu, C. Aimé, R. Gondo, T. Noguchi, N. Kimizuka, Angew. Chem. Int. Ed, 2009, 48, 9465-9468.